Note: Due to the number of Tweets embedded in this post the page may take some time to load.
Monday's sessions included the Gyn Cancer Poster session and the Poster Discussion session. There were a few other sessions that I found interesting and are included at the end of this post.
Poster Discussion Session
Initial efficacy and safety
results from ENGOT-ov60/GOG-3052/RAMP 201: A phase 2 study of
avutometinib (VS-6766) ± defactinib in recurrent low-grade serous
ovarian cancer (LGSOC). ABSTRACT 5515
🎯 The combination of avutometinib (RAF/MEK clamp) and defactenib (FAK inhibitor) demonstrates impressive activity in low grade serous #OvarianCancer, with higher efficacy in tumors that have RAS aberrations#ASCO23 #gyncsm pic.twitter.com/yArsGeAIxV
— Shannon Westin (@ShannonWestin) June 5, 2023
Randomized phase 2 study
of gemcitabine with or without ATR inhibitor berzosertib in
platinum-resistant ovarian cancer: Final overall survival (OS) and
biomarker analyses. ABSTRACT 5512
"Gemcitabine/berzosertib did not significantly improve OS versus
gemcitabine alone. Pts with PFI≤3 months and pts with RS-low tumors may
derive a survival advantage from addition of berzosertib to gemcitabine
in the platinum-resistant setting. Clinical trial information:
NCT02595892."
Correlation of cyclin E1
expression and clinical outcomes in a phase 1b dose-escalation study of
azenosertib (ZN-c3), a WEE1 inhibitor, in combination with chemotherapy
(CT) in patients (pts) with platinum-resistant or refractory (R/R)
epithelial ovarian, peritoneal, or fallopian tube cancer (EOC). ABSTRACT 5513
"Study assessed azenosertib + paclitaxel (PAC), carboplatin (Carbo), gemcitabine (GEM), or pegylated liposomal doxorubicin (PLD) in pts with metastatic high-grade serous EOC after ≤2 lines of chemotherapy. Patients with Cyclin E1 overexperessing tumors, a subgroup with suboptimal benefits from chemo , demonstrated significant imporovements in ORR and PFS vs patients with tumors having low expresssion."
A phase II trial of
palbociclib combined to letrozole after progression on second-line
chemotherapy for women with ER/PR-positive high-grade serous or
endometrioid ovarian, fallopian tube, or peritoneal cancer: LACOG 1018. ABSTRACT 5541
"Palbociclib combined to letrozole demonstrated a significant efficacy in
terms of PFS rate at 12 weeks (63.4%) and CBR (71.8%), with no new
safety concerns in women with recurrent advanced and metastatic hormone
receptor-positive ovarian cancer. These results warrant further
investigation of palbociclib plus letrozole in high-grade ovarian cancer."
The Normal Risk Ovarian Screening Study (NROSS): Twenty-one year update. ABSTRACT 5522
"A total of 7,856 healthy postmenopausal women were screened annually for a total of 50,596 women-years in a single arm study (NCT00539162). Serum CA125 was analyzed with the Risk of Ovarian Cancer Algorithm (ROCA) each year....An elevated ROCA, characterized by a significantly rising CA125, prompted referral of 2% of participants to transvaginal sonography (TVS) each year and required only 2 operations to detect each case of ovarian cancer, indicating that CA125 used in this way is adequately specific for effective screening.
Posters
Here are a few posters I found interesting that were not part of the discussion session.
Efficacy and
safety of niraparib maintenance therapy in patients with newly diagnosed
advanced ovarian cancer who had measurable residual disease: A post-hoc
subgroup analysis of the PRIME study. ABSTRACT 5562
"This study aims to report the efficacy, including antitumor activity, and safety of niraparib maintenance therapy in patients with measurable residual disease MRD after first-line platinum-based chemotherapy1LCT from the phase 3 PRIME trial. ... Median PFS (95% CI) was 22.3 (8.7–not estimable) months with niraparib versus 8.3 (5.6–11.0) months with placebo (hazard ratio, 0.36; 95% CI, 0.19–0.71)."
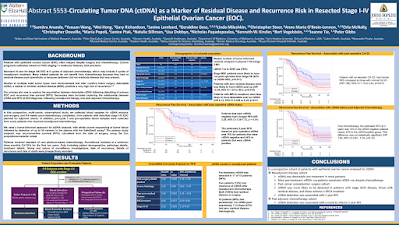
Circulating tumor DNA
(ctDNA) as a marker of residual disease and recurrence in resected stage
I-IV epithelial ovarian cancer (EOC). ABSTRACT 5553
Safety and efficacy of PIPAC in ovarian cancer patients with peritoneal metastases: A first-in-US phase I study. ABSTRACT 5554
"Pressurized intraperitoneal aerosolized chemotherapy (PIPAC) is a novel IP drug delivery method that optimizes tissue penetration depth and drug distribution, to treat recurrent peritoneal malignancies.
PIPAC with cisplatin/doxorubicin in platinum-resistant ovarian cancer is well tolerated. Intraperitoneal responses were seen in a subset of low-grade serous ovarian cancer patients, which may warrant further study."
Miscellaneous ASCO topics:
Patient Centered Research
Discussion by @DrDawnHershman
— Margaret Van Meter, MD (@MVanMeterMD) June 5, 2023
Patient-centered research allows:
*better decision making
*better outcomes
*better tolerability
*better dissemination
*longer time on drug
*better quality of life#ASCO23 #bcsm pic.twitter.com/36FbM3e37R
Patient centered research ! The theme of #ASCO23. It’s been fun being here in person! pic.twitter.com/OBFzJe1a27
— Julia Katherine (@jackel626) June 5, 2023
Solid tumor research
DESTINY PAN TUMOUR 02 study
— Yvette Drew (@YvetteDrew) June 5, 2023
🌟Great responses in GYN cancers🌟
Including #cervicalcancer ⬇️⬇️⬇️#ASCO23 #gyncsm https://t.co/fY8tBsUajn
Dee
Every Day is a Blessing !